Oral Semaglutide vs Empagliflozin: PIONEER 2 Study
- Ms. Tanvi Goel

- Apr 6
- 4 min read
Updated: Apr 7

Type 2 diabetes mellitus (T2DM) is a major global health challenge, disproportionately affecting low- and middle-income countries. In 2024, 589 million adults aged 20–79 years were living with diabetes, causing 3.4 million deaths and over US$1 trillion in health expenditure; prevalence is projected to reach 853 million by 2050 (1). Beyond hyperglycaemia, T2D is increasingly recognized as a complex, multisystem disorder associated with the cardiovascular, renal, and hepatic systems (2). Chronic hyperglycaemia, insulin resistance, and metabolic dysregulation collectively drive endothelial dysfunction, inflammation, and progressive organ damage, increasing the risk of cardiovascular disease, chronic kidney disease, and non-alcoholic fatty liver disease (3). Despite advances in therapeutic options, achieving durable glycaemic control alongside meaningful organ protection remains an unmet clinical need (4).
Metformin remains the recommended first-line therapy (5). However, it offers largely glucose-centric action, primarily targeting hepatic glucose production without directly addressing the broader pathophysiological drivers of organ damage (6). Consequently, many of the patients fail to achieve durable glycaemic control, and the residual cardiovascular and renal risk remains high (5). This gap necessitates therapies that not only lower glucose effectively but also address weight, cardiovascular risk, and long-term organ outcomes (5). In this evolving paradigm, drug classes such as sodium–glucose cotransporter-2 inhibitors (SGLT2i), such as empagliflozin, and glucagon-like peptide-1 receptor agonists (GLP-1 RAs), such as semaglutide, have gained prominence due to their pleiotropic benefits (7).
SGLT-2i reduces the renal reabsorption of glucose, increasing urinary glucose excretion (8). In contrast, GLP-1 RAs lower blood glucose while promoting weight loss, improving lipid profiles, and lowering blood pressure (9). Both classes significantly reduce cardiovascular risk and improve outcomes in T2D, making them valuable therapeutic options. Among these, semaglutide has shown robust glycaemic efficacy (10), meaningful weight reduction (11), and emerging benefits in cardiovascular, hepatic, and renal protection through anti-inflammatory.
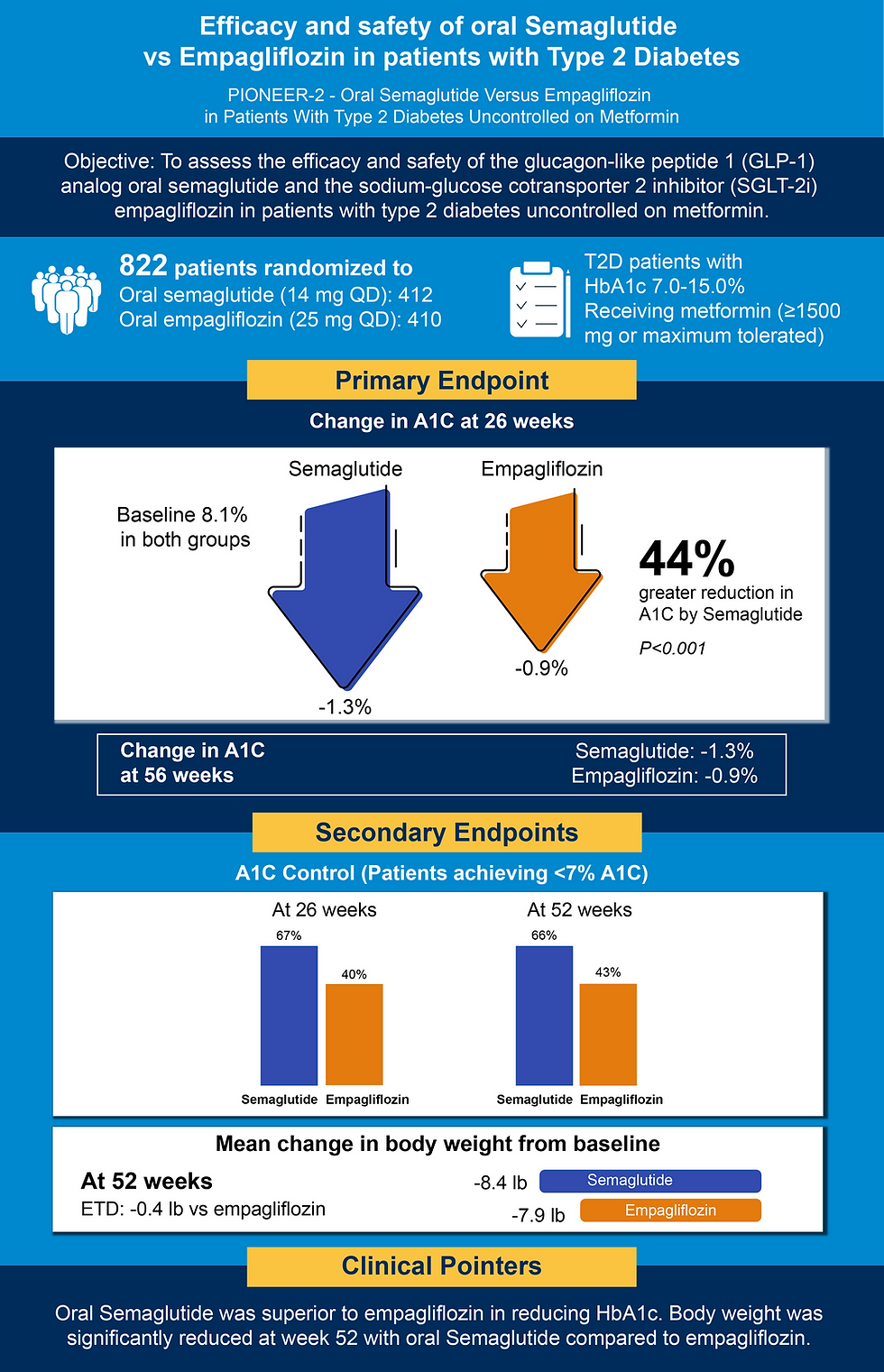
Within this context, the PIONEER 2 study by Rodbard HW et al. (12). provides comparative evidence between oral semaglutide and empagliflozin in patients with T2D inadequately controlled on metformin. This randomized trial evaluated HbA1c and body weight, key determinants of long-term metabolic and cardiovascular outcomes. Oral semaglutide showed superior HbA1c reduction, with more patients achieving glycaemic targets, along with meaningful weight loss. These findings indicate benefits beyond glucose lowering, supporting broader metabolic and organ protection effects.
Thus, PIONEER 2 underscores the need for therapies that address underlying disease mechanisms and systemic outcomes. As care shifts toward a cardiorenal-metabolic approach, semaglutide may help bridge the gap between glycaemic control and long-term organ preservation.
Study Summary
About the Author
Ms. Tanvi Goel is a Medical Writer with a master’s degree in pharmaceutical science. Tanvi has experience in scientific and regulatory writing and is eager to learn more about novel pharmacological agents.
Abbreviations
A1C: Glycated hemoglobin; ETD: estimated treatment difference; GLP - 1: Glucose like peptide 1; SGLT2i: Sodium-glucose cotransporter-2 inhibitors;
Reference
International Diabetes Federation. IDF Diabetes Atlas, 11th ed. Global factsheet (2025 update). 2025. Accessed April 3, 2026. https://diabetesatlas.org/
Zhu L, Chen C, Lu F, Li B. Systemic effects of type 2 diabetes therapies: an integrated perspective on the cardio–renal–cerebral–metabolic axis. Front Med (Lausanne). 2023;13:1746457.
Rohm TV, Meier DT, Olefsky JM, Donath MY. Inflammation in obesity, diabetes, and related disorders. Immunity. 2022;55(1):31-55.
Afeez A, Rose M. Emerging paradigms in type 2 diabetes management: beyond glycemic control with SGLT inhibition.
Wilcox T, De Block C, Schwartzbard AZ, Newman JD. Diabetic agents, from metformin to SGLT2 inhibitors and GLP1 receptor agonists: JACC focus seminar. Journal of the American College of Cardiology. 2020 Apr 28;75(16):1956-74.
Rena G, Hardie DG, Pearson ER. The mechanisms of action of metformin. Diabetologia. 2017;60(9):1577-1585.
Scheen AJ. GLP-1 receptor agonists and SGLT2 inhibitors in type 2 diabetes: pleiotropic cardiometabolic effects and add-on value of a combined therapy. Drugs. 2024;84(11).
Caruso I, Giorgino F. SGLT-2 inhibitors as cardio-renal protective agents. Metabolism. 2022;127:154937.
Meier JJ. GLP-1 receptor agonists for individualized treatment of type 2 diabetes mellitus. Nat Rev Endocrinol. 2012;8(12):728-742.
Aroda VR, Capehorn MS, Chaykin L, Frias JP, Lausvig NL, Macura S, Lüdemann J, Madsbad S, Rosenstock J, Tabak O, Tadayon S. Impact of baseline characteristics and beta-cell function on the efficacy and safety of subcutaneous once-weekly semaglutide: a patient-level pooled analysis of the SUSTAIN 1-5 trials. Diabetes Obes Metab. 2020;22(3):303-314.
Wharton S, Freitas P, Hjelmesæth J, Kabisch M, Kandler K, Lingvay I, Quiroga M, Rosenstock J, Garvey WT. Once-weekly semaglutide 7.2 mg in adults with obesity (STEP UP): a randomized controlled phase 3b trial. Lancet Diabetes Endocrinol. 2025;13(11):949-96
Rodbard HW, Rosenstock J, Canani LH, Deerochanawong C, et al.; PIONEER 2 Investigators. Oral Semaglutide Versus Empagliflozin in Patients With Type 2 Diabetes Uncontrolled on Metformin: The PIONEER 2 Trial. Diabetes Care. 2019 Dec;42(12):2272-2281.
Disclaimer
This content has been prepared by independent medical writers from diverse healthcare backgrounds, including members of the MedWriters Alumni Network. While every effort has been made to ensure accuracy, the authors, RxNews team, partners, agents, and sponsors accept no responsibility for any errors, omissions, or consequences arising from the use of this article. Mention or omission of any product does not imply endorsement or rejection.
For feedback or complaints, please contact us at contact@crixus.co.in.


Comments